Identify
To identify medicines uniquely and unambiguously, the Global Trade Item Number (GTIN) is used in combination with the batch number and the expiry date. For prescription medicines, it is mandatory to add a serial number. The latter to comply with FMD legislation.
Capture
This unique identification of medicine is applied to the product and/or packaging in the format of a barcode. This barcode will ensure that the medicine can be scanned throughout the logistics chain, which ends up with the patient.
Which barcode you use depends on the type of packaging in combination with what it is used for.
| WArehouse/pallet | Hospitals | Patient/hospital bed | Pharmacy | Retailer (over the counter)/ Point-of-sales | |
| Identify | GTIN-13 & GTIN-14 | GTIN-13 & GTIN-14 | GTIN-13 & GTIN-14 | GTIN-13 & GTIN-14 | GTIN-13 & GTIN-14 |
| Capture | GS1-128 | GS1-128 and GS1 Datamatrix | GS1-128 and GS1 Datamatrix | GS1 Datamatrix | EAN-13 |
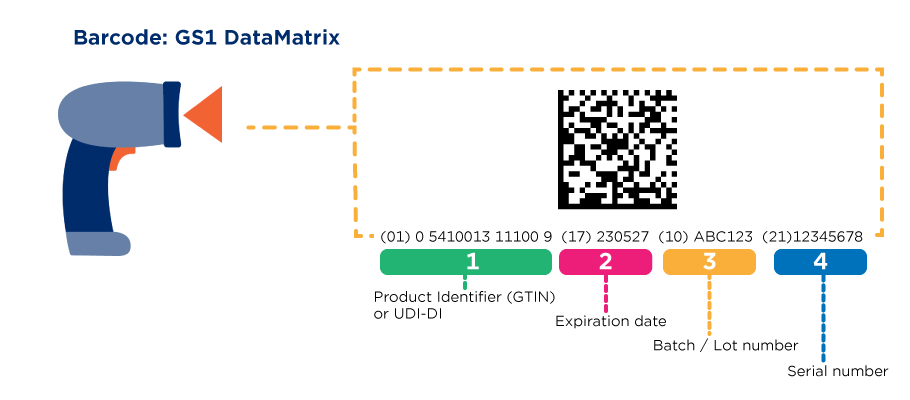
By means of Application Identifiers (AIs), this information is encoded in a GS1 Data Matrix or a GS1-128.
How to use Application Identifiers for medicines?
-
Global Trade Item Number – Application Identifier (01)
Is always represented 14 positions numerically. If it is e.g. a GTIN-13, a 0 is added on the left.
Examples:
- Product A: (01)05412345678908
- Product B: (01)05410013111009
-
Expiry Date – Application Identifier (17)
The expiry date has a fixed length and is always presented in the format 'YYMMDD'. If the expiry date is a month/year, the day may be represented by 00 until 1 January 2025. We suggest changing to the last day of the month as soon as possible.
Examples:
- 27 may 2023 : (17)230527
- 9 february 2020: (17)200209
- 30 june 2021: (17)210630
-
Batch- of lotnumber – Application Identifier (10)
A batch or lot number has a variable length and can contain up to 20 alphanumeric data.
Examples:
- (10)ABC123
- (10)N98756123-8
-
Serienumber – Application Identifier (21)
A serial number is of variable length and may contain up to 20 alphanumeric digits.
Examples:
- (21)12345678
- (21)AB1325456876123