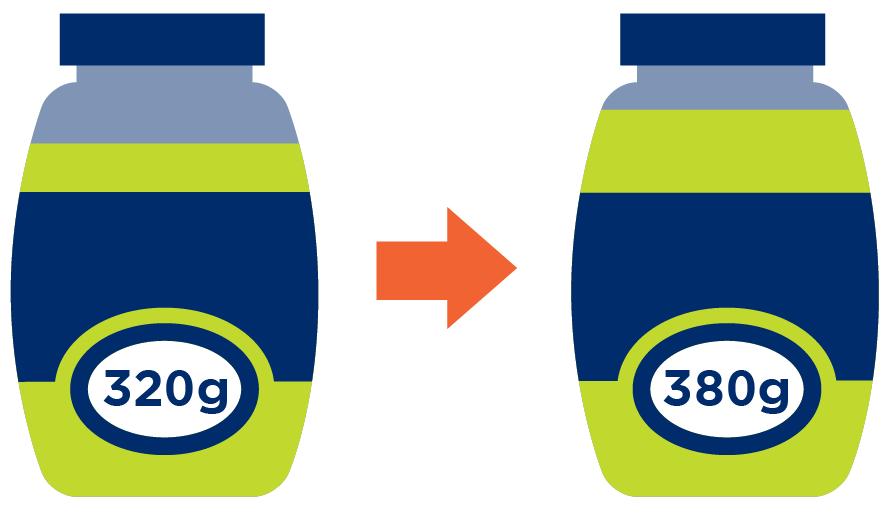
If certain characteristics of a product that is already being sold change, such as different dimensions or a new formula, the packaging may require a new Global Trade Item Number (GTIN). Whether this is the case depends on the type of change.
Within the supply chain, it has to be possible to distinguish between products before and after changes in certain cases.
The rules in this regard are laid down in the international GTIN Management Standard.
It contains realistic examples that help users make sound, consistent decisions.
Product identification in healthcare
Why correct product identification is so important in healthcare
In healthcare, a mistake in identifying medication or materials can be life-threatening. That’s why the sector uses GTINs to uniquely identify products.
What if a product changes?
Sometimes a new GTIN is needed – for example, when a new certification label is added to the packaging. The GTIN allocation rules help you decide when that’s required.
GS1 Healthcare developed an online decision tool. This way, you know exactly when you need to allocate a new GTIN.
